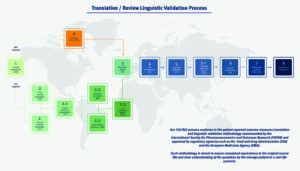
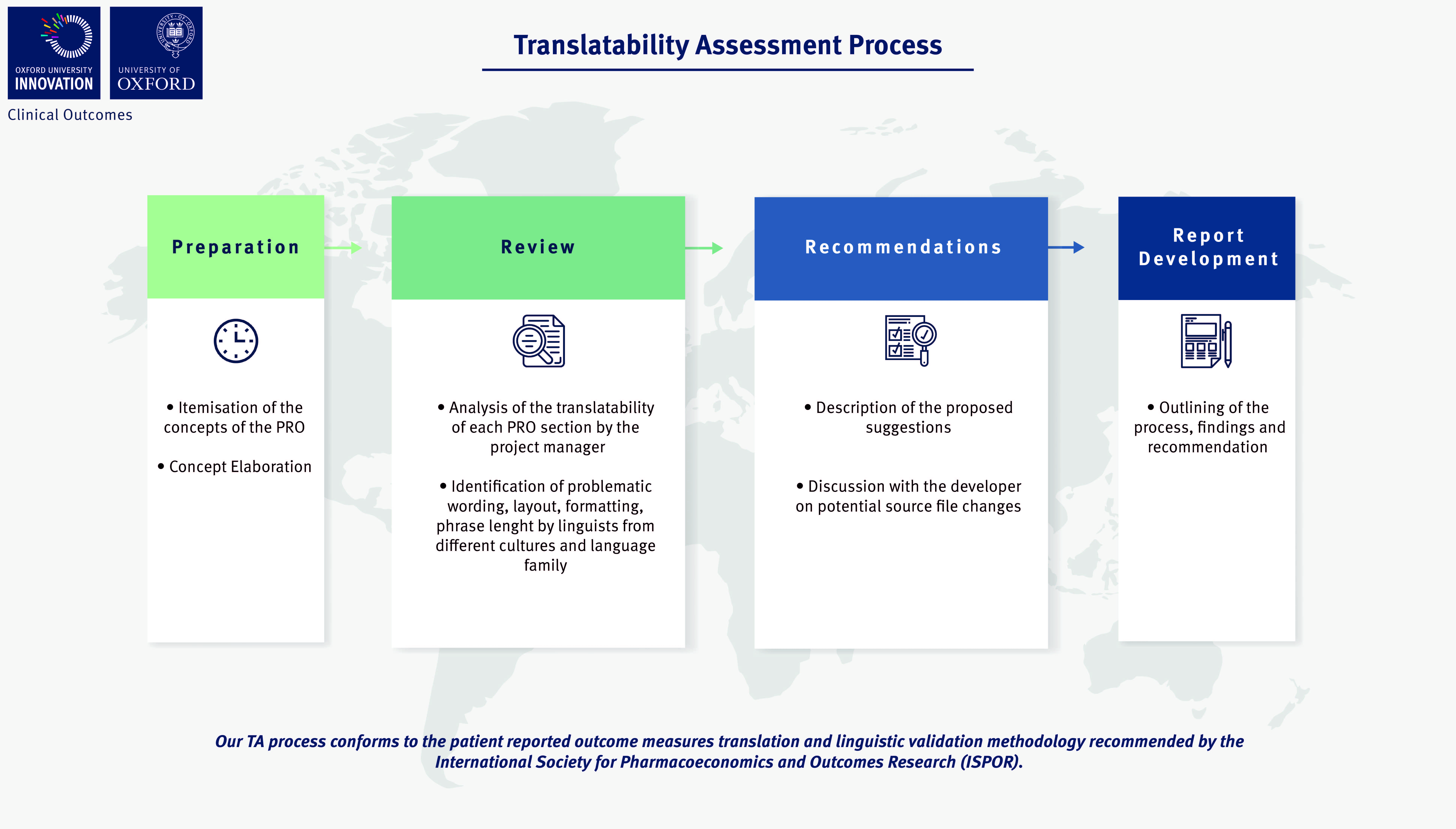
Our Translation/ Review and Linguistic Validation process conforms to the clinical outcome assessments translation and linguistic validation methodology recommended by the International Society for Pharmacoeconomics and Outcomes Research (ISPOR) and approved by regulatory agencies such as the Food and Drug Administration (FDA) and the European Medicines Agency (EMA). Such methodology is meant to ensure conceptual equivalence to the original (normally validated) source file and clear understanding of the questions by a population of the intended target respondents in a real-life scenario.
The linguistic validation process includes the following steps:
^ Back to top