AFIRM: Amyloid Formation and Inhibition Mechanism
Protein aggregation is associated with neurodegenerative diseases such as Alzheimer’s disease and Parkinson’s disease. Understanding this phenomenon is expected to provide insights into the pathology of these diseases.
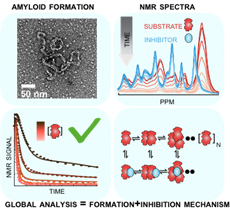
Oxford researchers have created an efficient software platform for the analysis of chemical kinetics of protein aggregation and chaperone mechanisms that makes use of nuclear magnetic resonance (NMR) data. This software enables accurate prediction of rate equation models, including determination of the rate and dissociation constants. The software has been tested with various datasets and a graphical user interface is being developed.
Amyloid mechanism
Many disorders including Alzheimer’s and Parkinson’s diseases are associated with amyloid formation and deposition in tissue. Improved understanding of the molecular mechanism of amyloid formation, and how this is perturbed by small molecules, proteins and chaperones is expected to support the discovery of diagnostic tests and therapeutics for such neurodegenerative diseases. Our method directly addresses this need.
Platform for mechanism determination
Oxford University researchers have developed an approach for mechanism determination. NMR spectroscopy can quantitatively follow the specific changes in aggregating molecules without the need or the addition of, for example, fluorescent tags. This method functions in the context of heterogeneous nonequilibrium mixtures such as inside cells. The approach provides experimental protocols for obtaining reproducible kinetic data, and globally analysing it to provide a molecular description of underlying events.
The AFIrM method has the following key features:
- Early events including the formation of primary nuclei can be identified and characterised.
- No normalisation of the data is required – this allows for a complete thermodynamic, as well as a kinetic description of the aggregation mechanism and inhibition mechanism.
- Method works in the presence of complex mixtures and in the presence of living cells.
- Software numerically efficiently evaluates rate equations allowing for exact testing of aggregation mechanisms of arbitrary complexity.
Commercialisation
Oxford University Innovation is interested in speaking to companies that would like to use our facilities as a service or license this software for in-house use.
about this technology

